ver the many years of my career, I have noticed that standards keep changing at an ever-increasing rate. Most recently, I have been involved in four of the standards committees dedicated to IEC 60601-1, Medical electrical equipment, one of eight standards in the IEC Amendments Project. Part of our work is reflected in the recently released Ed. 3.2 of IEC 60601-1.
Medical device standards are being developed more and more rapidly and some existing standards are being updated in shorter time frames (i.e., the rules for IEC standards development has changed to allow for shorter development cycles), and national medical device regulations (including guidances) keep changing at a faster pace. As a result, it is becoming more difficult for medical device manufacturers as well as medical device consultants to keep up to date with the proliferation of changes. Ultimately, this impacts the manufacturer’s quality systems and technical documentation, increases product development cycle times, and stretches out product time to market.
This article will focus on the IEC 60601 series of medical electrical standards, and specifically on the IEC Amendments Project, a project that was completed under Sub Committee 62A (SC62A). The article provides a summary of some of the changes from the previous version of the standards impacted by the Amendments. There are literally hundreds of changes in these standards, and it would be impossible to adequately provide details on all of these changes. But we’ll do our best in the pages that follow.
It is important to understand that the particular standards of IEC 60601-2-XX / IEC/ISO 80601-2-XX have not yet been updated to align with the Amendments. If the particular standard applicable to your device has not yet been updated to align with edition 3.2 of the general standard, you can continue to use edition 3.1. However, the new versions of collateral standards (e.g., IEC60601-1-2 ed 4.1) may still apply because of new regulatory requirements.
Some particular standards in the series are likely to be updated fairly quickly, while others may take up to three or more years before they are published. This extended timeline may determine when manufacturers begin the process of transitioning from IEC 60601-1, Ed. 3.1, and the applicable collateral and particular standards to the pertinent Amendments.
The decision to transition may be impacted by additional factors such as:
- Transition dates of national certifiers such as UL, CSA, BSI;
- National regulators transition periods;
- New product being ready for market or legacy product lines;
- Regulatory approvals;
- Existing safety certifications;
- Business, regulatory, quality system strategy and impact.
- Some of these changes were intended to align the standards with regulatory requirements and with the updates to ISO 14971, IEC 62366-1 and IEC 62304 to facilitate the regulatory approval process:
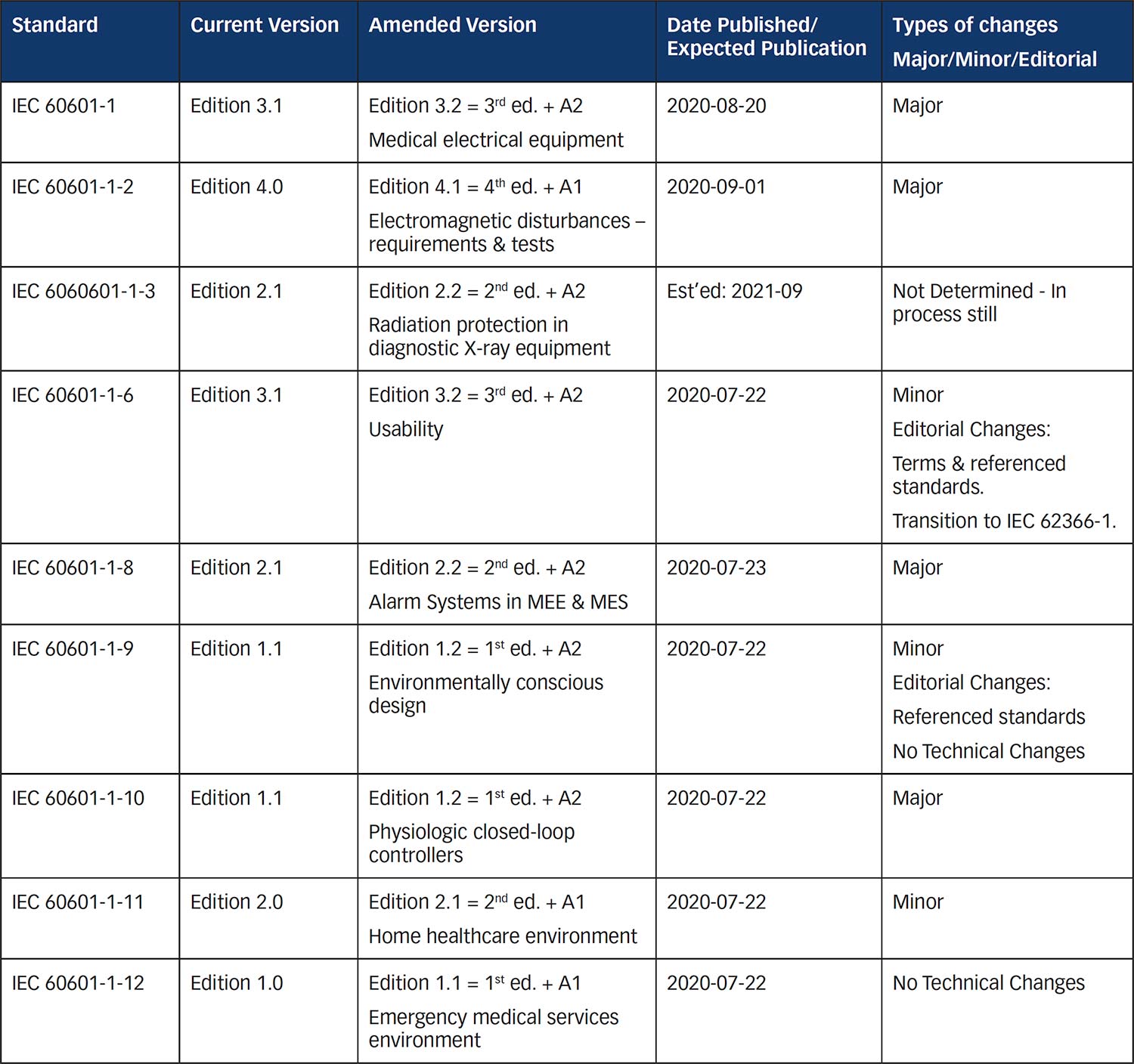
- IEC 60601-1, 60601-1-2, 60601-1-6 and 60601-1-10 refer to the most recent standard ISO 14971:2019 Medical devices – Application of risk management to medical devices standard.
- IEC 60601-1-6, 60601-1-8, 60601-1-10, &
60601-1-11 refer to the most recent standard IEC 62366-1:2015 + A1:2020 for Medical devices – Part 1: Application of usability engineering to medical devices. Note that IEC 60601-1 refers bibliographically to IEC 62366-1:2015 as an informative reference, not as a normative standard. - IEC 60601-1 refers to the current IEC 62304:2006 + A1:2015. It was hoped that IEC 62304 2nd edition would have been published but that edition had issues in committee and has not yet been published. So the Amendments Project couldn’t wait any longer to align with the anticipated IEC 62304 2nd edition requirements. We will have to live with this version for now.
- Updates to key standard references – Normative references that were updated in IEC 60601-1, Ed. 3.2 include the following standards (a number of which will be discussed later in this article):
- IEC 60601-1-2:2014 + A1:2020, EM disturbances
- IEC 60601-1-3:2008 + A1:2013, Diagnostic X-ray equipment
- IEC 60601-1-6:2010 + A1:2013 +A2:2020, Usability
- IEC 60601-1-8:2006 + A1:2012 + A2:2020, Alarm systems
- IEC 60747-5-5:2007 or later, Optoelectronic devices – Photocouplers
- IEC 60825-1:2014, Safety of laser products – Part 1: Equipment classification and requirements
- IEC 60950-1:2005 + A1:2009 +A2:2013, Information technology equipment
- IEC 62133-2, Lithium systems
- IEC 62368-1:2018, Audio/video, information and communication technology equipment
- ISO 7010:2019 Safety signs
- ISO 15223-1:2016, Medical devices— Symbols to be used with medical device labels, labelling and information to be supplied
- New or updated terms in IEC 60601-1 and some of the other standards. Some of the terms come from the regulatory standards. IEC 60601-1 has two new definitions internal to the standard itself.
- IEC 60601-1 required a significant number of clarifications, as did several other standards. The primary reason for these clarifications stemmed from:
- Safety gaps identified by WG14 decisions, many of which are published in IEC TR 60601-4-3:2018 (2nd Ed) Guidance and interpretation – Considerations of unaddressed safety aspects in the third edition of IEC 60601-1 and proposals for new requirements
- Inconsistencies within a standard
- Technical errors which generated new and updated test requirements
We found some drawbacks with IEC 62368-1:2018 when we did our analysis for an alternative option to IEC 60601-1. There are areas where voltages for 2 MOOP don’t meet the requirements for 1 MOPP, so manufacturers should carefully read and evaluate the examples and extensive details included in Clause 8 of Annex A (Guidance & Rationale) to determine if they apply to a given device or component, such as switch mode power supplies.
In many cases, working voltages that are above 354Vdc/250Vrms become problematic for double insulation for 2 MOOP for air clearance for IEC 62368-1:2018 as it may not necessarily meet the needed 1 MOPP for air clearance. Similar to IEC 62368-1, IEC60950-1:05, A1:09, A2:13 working voltages that are above 707Vdc/500Vrms in many cases become problematic for double insulation for 2 MOOP for air clearance as it may not necessarily meet the needed 1 MOPP for air clearance. Adding in IEC 62368-1 was not originally part of IEC 60601-1, Ed. 3.2 but was inserted into the project given the anticipated shortage of IEC 60950-1 certified power supplies in the near future.
If you can’t you use an IEC 62368-1 switch mode power supply, here are some other options:
- Substitute an IEC 60601-1 and IEC 60601-1-2 compliant power supply. This is our recommendation to clients anticipating FDA review and since reviewers may have concerns about the use of a power supply intended for ITE applications.
- Look at the isolation in your overall device/system and determine if you can add additional isolation that will get you the isolation needed. This may mean a redesign and additional testing, and could add cost and testing time.
The changes also relate to other components that provide MOOP isolation on the mains side of power isolation of medical devices, as well as system requirements related to monitors, keyboards, computers, printers, etc. The updates to IEC 60601-1, Ed. 3.2 reflect these considerations.

Figure 23 was the only one in the series of figures in the standard that had a 1 mm instead of X mm in the figure when the previous update was made. The X mm rules in Clause 8.9.4 had to be updated slightly to align properly but have been in the standard since Ed. 3.1.

New tests Table 11, Clause 8.11 immunity to proximity magnetic fields. Two of the three tests per Clause 8.11 (134.2 kHz @ 65A/m and 3.56Mhz @ 7.5A/m) are from the AIM 7351731 standard. The third test (30kHz @ 8A/m) is for the home healthcare environment (radiant cooktops).
The Guidance section on the application of risk management with regard to electromagnetic disturbances has been totally rewritten to clarify risk management references in the standard.
Clause 6.3.3.2 – The test set-up and configuration has been changed to correct references to figures and tables in ISO 3744. This means test source and locations (based on figures) will change. Therefore, the test results may vary from past results.
Added new distributed Alarm systems requirements in Clause 6.11.1.
What is not clear is how long it will take the FDA to “recognize” the particular standards (IEC 60601-2-XX & IEC/ISO 80601-2-XX) once they are aligned with the Amendments Project. I recommend that device manufacturers take a “state of the art” approach and apply the latest version of each standard when designing their devices, recognizing at the same time that this approach has limitations in cases where regulatory authorities have requirements that reference earlier editions of a given standard (i.e., MDD Harmonized Standards) and insist on using these outdated standards.
The goal of the Amendments Project was to make the more immediately needed changes to the IEC 60601-1 series of standards in advance of efforts to develop a 4th Edition of the standard, expected to begin by about 2025. We believe that the work of the Amendments Project will help clarify many important issues around the current use of IEC 60601-1 and its collateral standards, and make it easier to use the standard in the near term. At the same time, the changes are likely to result in some additional work, as device manufacturers will need to conduct a gap assessment and review their documents and systems to determine what needs to be updated before they resubmit to their test laboratories and regulators to meet these revised requirements.
